
HbA1c Testing at a GP Surgery: A Case Study on Improving Patient Pathway and QOF Compliance
Background
A Primary Care Network (PCN) has trialled measuring HbA1c on the QuikRead go Point of Care device in the diabetes clinic to optimise the management of diabetic patients.
There are 4.7 million people in the UK with Type 2 Diabetes. This number is predicted to increase to 5.5 million by 2030[1]. This places a significant burden on GPs to manage these patients.
Mismanagement of diabetes can lead to complications, with the number of lower limb amputations increasing year on year.
Every hour, someone with diabetes has a toe, foot or leg amputated.[2]
The trial led by a GP and Local Medical Committee Executive based at one of the GP surgeries within the PCN, used the QuikRead go device to measure HbA1c on all patients arriving at the Diabetes Clinic over the course of a one month period starting in June 2022.
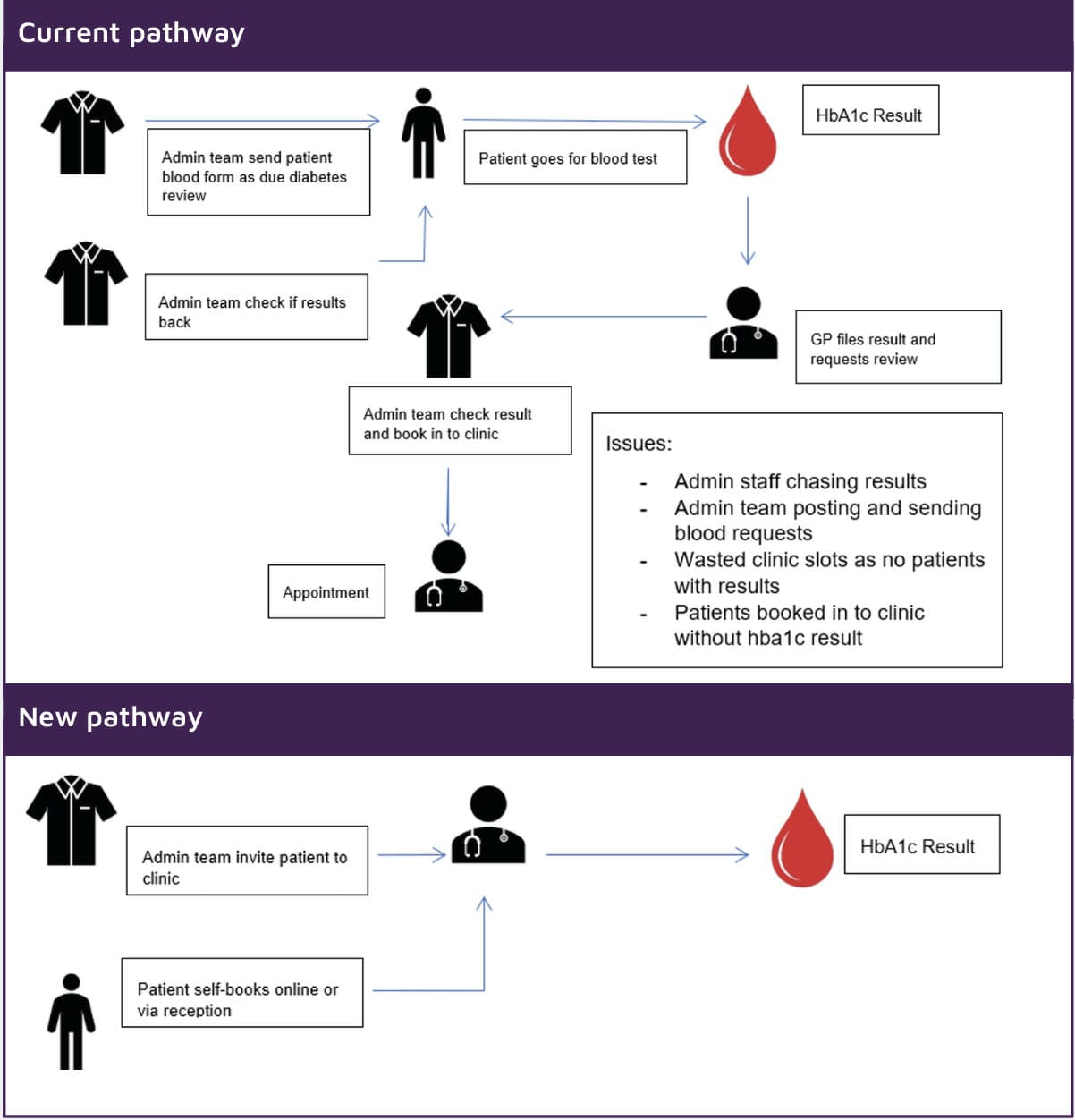
The aim was to improve the patient experience, reduce repeat appointments and reduce the administrative burden associated with diabetes check-ups.
Problems and inefficiencies that can arise when trying to schedule patients in for a Diabetes review range from:
- Administrative burden in arranging appointments
- Missed or repeat appointments due to blood results not being available
- Difficulties in providing appropriate and timely prescriptions without an HbA1c result
Along with this, there are Quality Outcome Framework (QOF) points dedicated to Diabetes; therefore, not only can point-of-care testing of HbA1c improve patient pathways, but it can also help GPs meet their QOF targets.
For the study, the diabetes nurse performed an HbA1c test on every diabetic patient attending the clinic for a review. The study involved 66 patients over the course of a one-month period. These results were then compared to the laboratory results.
Good correlation between the QuikRead go results and laboratory results was seen [Figure 1.]

The QuikRead go device uses 1ul of whole capillary blood and gives a result in five minutes. This allows for an HbA1c measurement to be taken during the check-up appointment, enabling the Diabetes nurse to make decisions on prescriptions and treatment plan.
The operational benefits offered by HbA1c testing in clinic were significant and a business case has been approved for the surgery to purchase a QuikRead Go to measure HbA1c in the diabetes clinic.
Figure 2 highlights the pathway improvements seen with the use of point-of-care HbA1c testing.
For point-of-care testing in blood diagnostics, Quality, Compliance and Connectivity are all points that need to be considered.
We at Una Health offer support to our customers by providing Quality Control material. We can support and recommend External Quality Assurance schemes and IT connectivity solutions.
With point of care testing becoming more widespread, Una Health can support you to establish your service, improve patient pathways and create a business case. Please enquire further here.
National Guidelines[3] recommend that all diagnostic pathology point of care testing equipment is registered with an External Quality Assessment scheme. The QPoint scheme is designed to assure healthcare professionals of the quality of their results.
You can see below the performance against a reference laboratory using the Adams method (HPLC).


Case study available for download:
HbA1c Testing at a GP Surgery: A Case Study on Improving Patient Pathway and QOF Compliance
References:
[1] Diabetes UK – https://www.diabetes.org.uk/about_us/news/new-stats-people-living-with-diabetes#:~:text=Diabetes%20prevalence,of%20those%20with%20type%202
[2] Diabetes UK – https://www.diabetes.org.uk/be-in-the-know-amputations
[3] MHRA Management and use of IVD Point of Care Test Devices v1.1 (2013)

Find out more – QuikRead go HbA1c
